
This paper is complementary to prior work where classroom activities were developed to provide insight into the statistical nature of entropy. These simple and quick calculations help students become familiar and comfortable with the concept of entropy. This paper describes a calculation of the increase in entropy that can be performed while completing three traditional thermodynamics experiments. 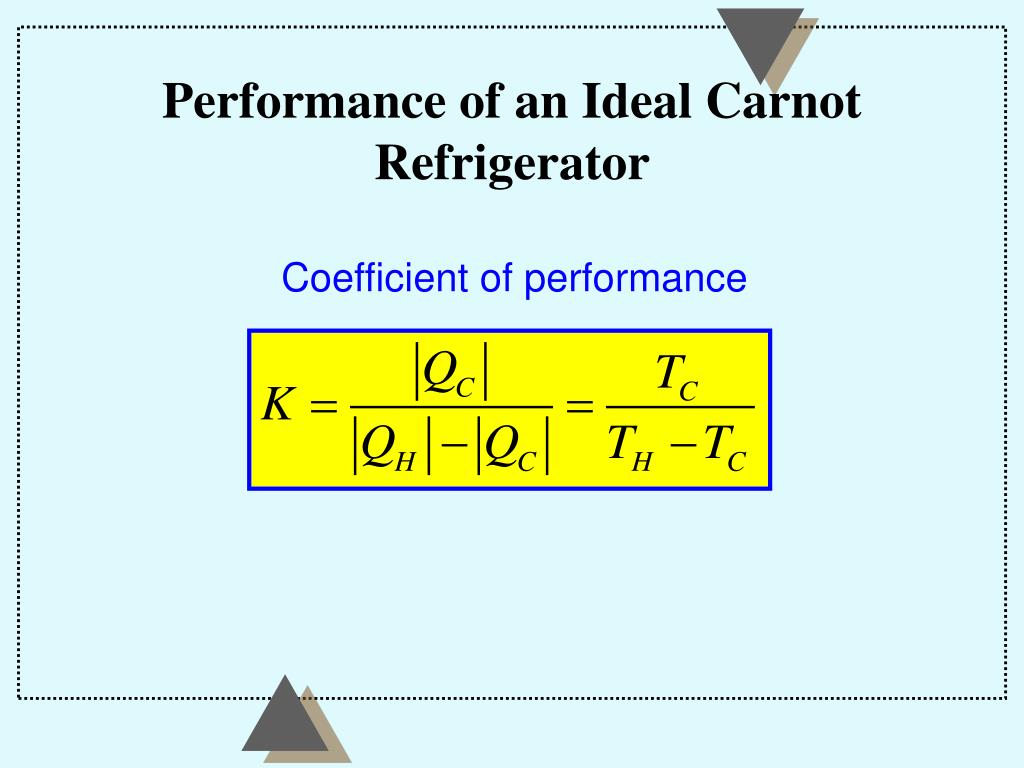
However, when the second law of thermodynamics is expressed using the concept of entropy (?S = 0, for a closed system), most first-year physics students lack any direct experimental experience with this parameter. The test begins with the definition that if an amount of heat Q flows into a heat reservoir at constant temperature T, then its entropy S increases by S Q / T. every natural system will tend toward lower entropy heat lost by one object must be gained. The concept of entropy was first introduced in 1850 by Clausius as a precise mathematical way of testing whether the second law of thermodynamics is violated by a particular process. Both these statements can be presented to physics students in a conceptual manner, and students' "everyday" experiences will support either statement of the second law of thermodynamics. heat always flows spontaneously from a colder body to a hotter one. There is the "Clausius formulation," which can be stated in a very intuitive way: "No process is possible whose sole result is the transfer of heat from a cooler to a hotter body." There is also the "Kelvin-Plank principle," which states that "no cyclic process exists whose sole result is the absorption of heat from a reservoir and the conversion of all this heat into work" (since this would require perfect energy conversion efficiency). The second law of thermodynamics has various formulations.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
